
The collision and the repulsion cause the alpha particle to “bounce” backwards and move on a very different path. What causes the alpha particles to deflect backwards?Ī tiny number of alpha particles, traveling at 10% of the speed of light, hit a dense atomic center right in its middle. In this experiment, Rutherford tested Thomson’s Plum Pudding Model by attempting to pass a beam of alpha particles through a thin gold foil. What disproved plum pudding model?Īnswer and Explanation: Rutherford disproved the Plum Pudding Model of the atom by conducting his gold foil experiment. The electrons revolve in circular orbits about a massive positive charge at the centre. Rutherford proposed that the atom is mostly empty space. 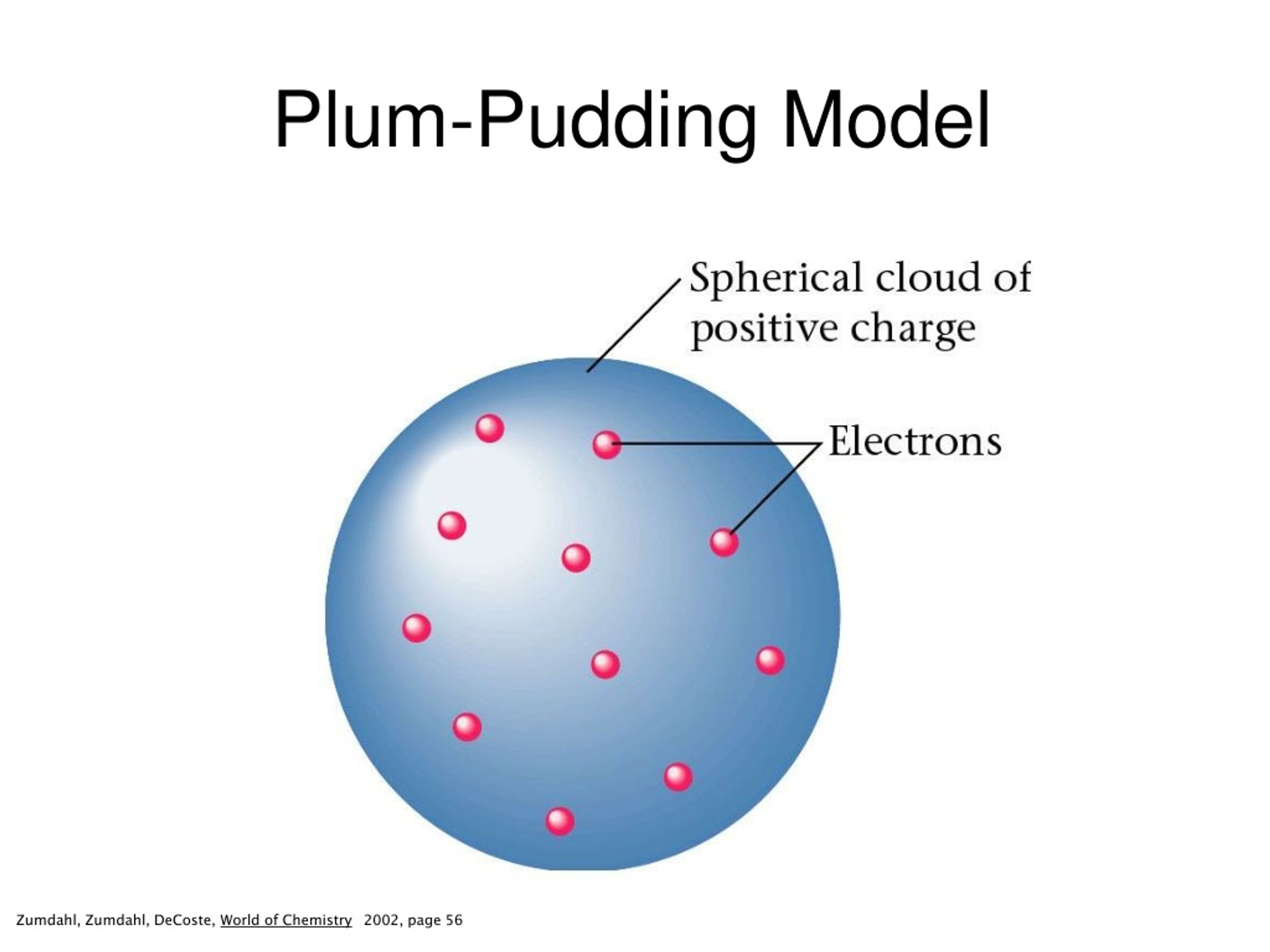
The symmetrical distribution of charge would allow all the α particles to pass through with no deflection. He argued that the plum pudding model was incorrect. The plum pudding model is defined by electrons surrounded by a volume of positive charge, like negatively-charged “plums” embedded in a positively-charged “pudding” (hence the name). The plum pudding model (also known as Thomson’s plum pudding model) is a historical scientific models of the atom. Although most alpha particles passed straight through, some were scattered at large angles, or even came back. We’ve been looking at how Ernest Rutherford showed that Thomson’s plum pudding (think of Christmas pudding or fruit cake) model of the atom was incorrect by firing alpha particles at a piece of thin gold foil. Why was the plum pudding experiment inaccurate? This effectively disproved the notion that the hydrogen atom was the smallest unit of matter, and Thompson went further to suggest that atoms were divisible. 
Upon measuring the mass-to-charge ration of these particles, he discovered that they were 1ooo times smaller and 1800 times lighter than hydrogen. Thomson proposed the plum pudding model of the atom, which had negatively-charged electrons embedded within a positively-charged “soup.” How JJ Thomson’s plum pudding model disproved Dalton’s atomic theory? Thomson’s experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. 24 Why was Thomson’s model not accepted? What did the plum pudding experiment prove?
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
